Beer's Law and Beyond
Keith B. Oldham and J. Mark Parnis
Chemical Properties Research Group, Department of Chemistry, Trent University, Peterborough, Ontario, Canada.
Beer’s law describes the diminution of intensity as light traverses an absorbing medium.
1 1. The classical law and its history
When electromagnetic radiation, such as ultraviolet or visible light, passes through a transparent medium that contains an absorber of that illumination, the radiation’s intensity diminishes steadily with passage through the medium. Commonly the radiation is in the form of a collimated beam that impinges perpendicularly on a slab of width [math]L[/math] of the medium, as suggested diagrammatically in Figure 1. One may conjecture that, at any illuminated plane [math]x[/math] within the medium, the decrease in the intensity [math]I[/math] of the radiation with distance would be proportional to the uniform concentration [math]c[/math] of the absorber and to the local intensity of the light at that point; that is
[math] \frac{\mathrm d}{\mathrm d x} I(x)=-\alpha cI(x)\tag {1} [/math]
where [math]\alpha[/math] is a proportionality constant. Integration of this equation leads to
[math] \ln\frac{I(0)}{I(x)}=\alpha c x\tag{2} [/math]
whence, on choosing [math]x[/math] to be the exit plane for the radiation, and with [math]\varepsilon=\alpha ln(10)=2.303\alpha[/math]:
[math] \log_{10}\frac{I(0)}{I(L)}=\varepsilon c L\tag {3} [/math]
Citing the earlier findings [1] of Pierre Bouguer, the 1760 treatise [2] of Johann Lambert publicized the linear dependence of the logarithm of the [math]I(0)/I(L)[/math] ratio on [math]L[/math], whereas its analogous dependence on [math]c[/math] remained unrecognized until the work [3] of August Beer almost a century later. Equation (3) is the form in which Beer’s law (also known as the Beer-Lambert or Beer-Lambert-Bouguer law) is most commonly encountered.
The constant [math]\varepsilon[/math] goes by a variety of names, of which absorptivity, extinction coefficient, absorbancy index and attenuation coefficientare four. Frequently and confusingly, those names, or similar ones, are also given to; [math]\alpha[/math] moreover, the symbol [math]\varepsilon[/math] often replaces [math]\alpha[/math] in equation (2). Further misperception can arise because [math]\varepsilon[/math] has the inverse units of (concentration) [math]\times[/math] (distance) and its numerical value therefore reflects the unit chosen for concentration, which variously may be moles per litre, molecules per cubic metre or one of several kinds of percentage. Absorptivity is generally a function of wavelength and therefore Beer’s law is strictly valid only if the radiation is monochromatic. Furthermore, the law fails at high concentrations or if there is significant light scattering from turbidity.
Any of equations (1), (2) or (3), as well as equations (4) and (5) below, may be said to be mathematical expressions of Beer’s law. A verbal statement of the law would be:
When radiation traverses a medium containing an absorbing but photostable component, the decrease of radiant intensity with distance, at any point in the medium, is proportional to the concentration of the absorber and to the local light intensity, and therefore the radiant intensity varies exponentially with distance.
2 2. Photometry
The reciprocal of the [math]I(0)/I(L)[/math] ratio is known as the transmittance [math]T[/math], whereas the decadic logarithm of this ratio is defined as the absorbance, [math]A[/math]; accordingly
[math] T=\frac{I(L)}{I(0)}10^{-\varepsilon c L}\tag {4} [/math]
and
[math] A=log_{10}\frac{I(0)}{I(L)}=\varepsilon c L\tag {5} [/math]
Both these quantities are dimensionless and therefore independent of the units (of which there are many alternatives) in which the light intensity is measured. Alternative names for [math]A[/math] are absorbancy and optical density and one-tenth of this quantity has been called the transmission loss (in decibels).
The primary usage of Beer’s law in chemistry is in measuring concentrations via photometry or spectrophotometry [4]. The “spectro” in the latter name reflects the frequent utility of measuring absorbance as a function of the radiation’s wavelength, thereby generating an absorption spectrum. The medium is most often a liquid solution housed in a cuvette (a rectangular “box” of standard interior length, with transparent window-like walls); but gaseous and solid media are investigated similarly. Figure 1 resembles a schematic diagram of a spectrophotometer. The intensity of the monochromatic light exiting the medium is measured and compared either with the incident intensity at [math]x=0[/math], or with the emergent intensity from the same or a matched cuvette with the absorber absent. The output of the instrument is generally in the form of a transmittance and/or an absorbance measurement. A standardizing experiment is also performed, using a solution of known absorber concentration, whereby [math]\varepsilon[/math] is determined or confirmed. In photometry, the concentration of absorber in the cuvette is generally uniform and unchanging, but a uniform concentration is not mandatory. Beer’s law holds whether or not the concentration is uniform, because the solution to the differential equation
[math] \frac{\mathrm d}{\mathrm d x} I(x)=-\alpha c(x)I(x)\tag {6} [/math]
is given by a version of equation (3) in which the [math]“c”[/math] in that equation is replaced by the average concentration along the light path, so that
[math] A=log_{10}\frac{I(0)}{I(L)}=\varepsilon \bar{c} L \;where\; \bar{c}=\frac{1}{L}\int\limits_0^Lc(x)dx\tag {7} [/math]
This conclusion is easily rationalized because it is the number of light-absorbing molecules, and not their distribution, that is crucial in abbreviating the light intensity.
3 3. The bivariate Beer laws
A spatial variability of concentration can arise from the illumination itself if the light-absorbing molecules, on capturing the light’s photons, suffer a chemical change. Typically, only a small fraction of photon absorptions lead to chemical transformation, most photons being absorbed harmlessly.Not only does the absorber concentration become a temporal as well as a spatial variable but, in consequence, the light intensity is forced to follow suit, becoming perturbed away from the exponential spatial dependence predicted classically. Time, which has no relevance to the classical Beer law, becomes a player. Provided the decomposition product does not itself emit or absorb light, the partial derivative
[math] \frac{\partial }{\partial x} I(x,t)=-\alpha c(x,t)I(x,t)\tag {8} [/math]
replaces equation (1). Because it involves two independent variables – distance and time – equation(8) is named the bivariate Beer intensity law. The rate of light-induced decomposition would be expected to be proportional both to the local light intensity and to the concentration of absorber, and this expectation is generally realized. Accordingly, the equation,
[math] \frac{\partial }{\partial t} c(x,t)=-\beta I(x,t)c(x,t)\tag {9} [/math]
named the bivariate Beer concentration law, describes the local rate of chemical change, [math]\beta[/math] being a proportionality constant. Notice the similarities between equations (9) and (8): in form, the dependence of concentration on time exactly matches the dependence of light intensity on distance. A single function must describe both! The identity of that function was recently established [5]; it is called the bleaching function in recognition of the prototype problem to which it applies being the bleaching, by light, of dye solutions. The bleaching function solves the problem posed by the twin differential equations (9) and (8), subject to the boundary conditions [math]c(x,0)=c_{0}[/math] a constant, and [math]I(0,t)=I_{0}[/math], another constant; thus
[math] c(x,t)=c_{0}\exp(-\beta I_{0}t)\displaystyle\sum_{k=0}^{\infty}\frac{(-\alpha c_0x)!}{k!}B_k\{\exp(-\beta I_0t)\} [/math] [math]\tag {10}[/math] [math] I(x,t)=I_{0}\exp(-\alpha c_{0}t)\displaystyle\sum_{k=0}^{\infty}\frac{(-\alpha I_0t)!}{k!}B_k\{\exp(-\alpha c_0x)\} [/math]
The terms [math]B_{k}[/math] are members of an infinite set of polynomial functions of which the first few members are
[math] B_{0}(z)=1\\ B_{1}(z)=z-1\\ B_{2}(z)=2z^2-3z+1\\ \dots\\ B_{k}(z)=\sum_{h=0}^{k}b_{k, h}z^{k}\tag {11} [/math]
The recursion relationship [math] B_{k+1}(z)=[z-1]d(zB_{k} (z))/dz[/math] applies but, for computer implementation, the recurrence of the coefficients,
[math] b_{k+1, h}= hb_{k, h-1}-(h+1)b_{k, h}\tag {12} [/math]
is more useful.
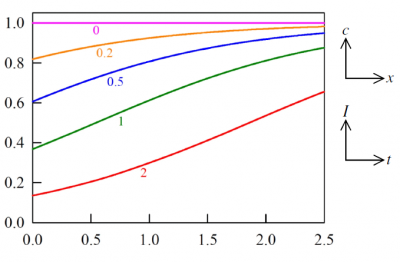
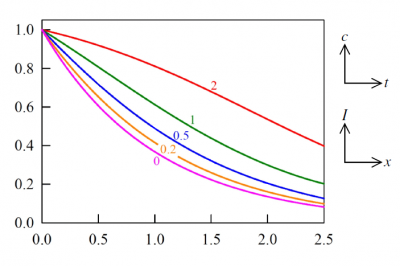
Figures 3 and 4 show typical profiles of absorber concentration and light intensity during an experiment governed by equations (9) and (8). The duality in the interpretation of these diagrams underwrites the essence of the bivariate Beer laws, which could be stated in words as;
When radiation traverses a medium containing an absorbing and photolabile component, the decrease of radiant intensity with distance, at any point in space and at any time instant, is proportional to the rate of concentration decay with time. Concomitantly, the concentration increase with distance mirrors the growth in local light intensity with time.
Figure 3. Profiles calculated via equations (10). Axis labels are deliberately omitted because the figure may be interpreted in the two ways, as suggested by the small diagrams. Thus, one interpretation is that the curves are plots of concentration versus distance, initially and at four subsequent times. Alternatively, each curve may be regarded as depicting the strengthening light intensity, with time, at the entry plane and at a sequence of four other locations. Specifically, the plots are of [math]c(x,t)/c_{0}[/math] versus [math]\alpha c_{0}[/math] at the five listed values of [math]\beta I_{0}t[/math]; or equivalently they are plots of [math]I(x,t)/I_{0}[/math]versus [math]\beta I_{0}t[/math] for the five distinct values [math]\alpha c_{0}x=[/math]0, 0.2, 0.5, 1.0 and 2.o.
With “ein” abbreviating the “einstein” unit, (the number of photons divided by Avogadro’s number, or “a mole of photons”), a set of mutually consistent units for the quantities in the bivariate Beer laws is [math]x (m)[/math], [math]t (s)[/math], [math]c (mol \times m^{-3})[/math], [math]I(ein \times m \times s^{-1})[/math] and when expressed in the [math]{(ein \times m^{-2} \times s^{-1})}[/math] unit, light intensity is termed irradiance.
4 4. Photochemistry
Most often, when a photon is absorbed and annihilated by a molecule, the photon’s energy is dissipated as heat. Occasionally, however, the energy localizes in, and severs, one of the molecule’s bonds. This results in a photochemical reaction: either the rearrangement of the molecule or its disintegration into fragments. A third possibility is the emission of another photon, but here we discount that third outcome.
The fraction of absorbed photons that leads to chemical change is known as the quantum yield; it is a key factor in photochemical studies [6, 7]. For a specified photochemical process, the quantum yield is represented by the symbol [math]\varphi[/math], is expressed in units of [math]mol \times ein^{-1}[/math], and is treated as a constant for light of a given wavelength.
Imagine a cuvette of length [math]L[/math] and cross-sectional area [math]S[/math] housing a solution of a photolabile compound under irradiation. Consider the length to be subdivided into wafers of width [math]dx[/math]. The number of photons absorbed within a particular wafer in a time interval [math]dt[/math] is
[math] \text {photon lost}=S[I(x,t)-I(x+dx,t]dt=-S\frac{\partial I(x,t)}{\partial x}dxdt=\alpha Sc(x,t)I(x,t)dxdt\tag {13} [/math]
whereas the number of molecules destroyed within the wafer in that time interval is
[math] \text {molecules lost}=S[c(x,t)-c(x,t+dt]dx=-S\frac{\partial c(x,t)}{\partial t}dxdt=\beta Sc(x,t)I(x,t)dxdt\tag {14} [/math]
The final equalities in (13) and (14) come from incorporating equations (8) and (9) respectively. By definition, the ratio of the number given by expression (14) to that given by expression (13) is the quantum yield. On dividing the two formulas, the simple result
[math]\varphi=\frac{\beta}{\alpha}\tag {15}[/math]
emerges. This opens a route to the evaluation of quantum yield via photometry, a route that does not require knowledge of [math]\varepsilon[/math].
Though the two independent variables – distance and time – play analogous roles in the mathematics of the bivariate Beer laws, there are ways in which time is superior in an experimental setting. Absorbance can easily be followed as a function of time, but standard photometric instrumentation does not allow the distance dimension to be scanned in a comparable way. Moreover, the distance scale is limited to [math]0\leq{x}\leq{L}[/math], whereas time is limitless. Hence, studying the time dependence of the absorbance of a solution of a photolabile species can provide, by proxy, information on concentration and thereby on the quantum yield of the photochemical reaction. It has been demonstrated [8] that a plot versus time of the quantity
[math] \frac{A(T)}{A(0)}+\frac{1}{A(0)}ln\frac{1-T(t)}{1-T(0)}\tag {16} [/math]
in which both transmittance and absorbance appear, generates a linear graph of unity intercept and a slope of magnitude [math]\frac{-\varphi I_0}{c_0L}[/math]. In this way, Beer’s law can yield an important photochemical parameter.
Following a protracted birth, Beer’s law continues to mature and develop.
5 References
[1] Bouguer, Pierre. 1729. Essai d’optique sur la graduation de la lumiere.Paris: Jombert.
[2] Lambert, Johann H.1760. Photometriasive de mensura et gradibusluminis, colorum et umbraeAugsberg: Klett.
[3] Beer, August. 1852.“Bestimmung der Adsorption des rothen Lichtinfarbien Flussikeiten.” Annalen der Physik und Chemie 8674-88.
[4] Gore, Michael G, editor. 2000. Spectrophotometry and spectrofluorimetry: a practical approach. Oxford and New York: Oxford University Press.
[5] Oldham, Keith B. 2014.“The propagation of radiation through a medium containing a component that absorbs the radiation and is steadily destroyed by it.”Journal of Mathematical Chemistry 52 1007-1019.
[6] Wardle, Brian. 2009. Principles and Applications of Photochemistry. Chichester: Wiley.
[7] Klán, Petr, and Jakob Wirz. 2009. Photochemistry of organic compounds: from concepts to practice. Chichester: Wiley.
[8] Parnis, J Mark, and Keith B.Oldham. 2013. “Beyond the Beer-Lambert law: the dependence of absorbance on time in photochemistry.” Journal of Photochemistry and Photobiology, A: Chemistry 2676-10.